- Back to Home »
- farmasi »
- Potassium Hydroxide/Potasium hidroksida
Posted by : andry natanel
1 Nonproprietary Names
BP: Potassium Hydroxide
JP: Potassium Hydroxide
PhEur: Potassium Hydroxide
USP-NF: Potassium Hydroxide
2 Synonyms
Caustic potash; E525; kalii hydroxidum; kalium hydroxydatum;
potash lye; potassium hydrate.
3 Chemical Name and CAS Registry Number
Potassium hydroxide [1310-58-3]
4 Empirical Formula and Molecular Weight
KOH 56.11
5 Structural Formula
See Section 4.
6 Functional Category
Alkalizing agent.
7 Applications in Pharmaceutical Formulation or Technology
Potassium hydroxide is widely used in pharmaceutical formulations to adjust the pH of solutions. It can also be used to react with weak acids to form salts. Therapeutically, potassium hydroxide is used in various dermatological applications. 8 Description Potassium hydroxide occurs as a white or nearly white fused mass. It is available in small pellets, flakes, sticks and other shapes or forms. It is hard and brittle and shows a crystalline fracture. Potassium hydroxide is hygroscopic and deliquescent; on exposure to air, it rapidly absorbs carbon dioxide and water with the formation of potassium carbonate.
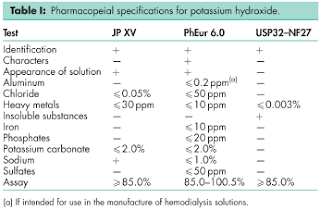
9 Pharmacopeial Specifications
See Table I.
10 Typical Properties
Acidity/alkalinity pH = 13.5 (0.1 M aqueous solution)
Melting point 360C; 380C when anhydrous
Solubility see Table II.
11 Stability and Storage Conditions
Potassium hydroxide should be stored in an airtight, nonmetallic container in a cool, dry place.
12 Incompatibilities
Potassium hydroxide is a strong base and is incompatible with any compound that readily undergoes hydrolysis or oxidation. It should not be stored in glass or aluminum containers, and will react with acids, esters, and ethers, especially in aqueous solution.
13 Method of Manufacture
Potassium hydroxide is made by the electrolysis of potassium chloride. Commercial grades may contain chlorides as well as other impurities.
14 Safety
Potassium hydroxide is widely used in the pharmaceutical and food industries and is generally regarded as a nontoxic material at low concentrations. At high concentrations it is a corrosive irritant to the skin, eyes, and mucous membranes. LD 50 (rat, oral): 0.273 g/kg (1)
15 Handling Precautions
Potassium hydroxide is a corrosive irritant to the skin, eyes, and mucous membranes. The solid and solutions cause burns, often with deep ulceration. It is very toxic on ingestion and harmful on inhalation. Observe normal handling precautions appropriate to the quantity and concentration of material handled. Gloves, eye protection, respirator, and other protective clothing should be worn. Potassium hydroxide is strongly exothermic when dissolved in ethanol (95%) or water, and considerable heat is generated. The reaction between potassium hydroxide solutions and acids is also
strongly exothermic. In the UK, the workplace exposure limit for potassium hydroxide has been set at 2 mg/m3 short-term.
16 Regulatory Status
GRAS listed. Accepted for use in Europe in certain food
applications. Included in the FDA Inactive Ingredients Database
(injections, infusions, and oral capsules and solutions). Included in
nonparenteral and parenteral medicines licensed in the UK.
Included in the Canadian List of Acceptable Non-medicinal
Ingredients.
17 Related Substances
Sodium hydroxide.
18 Comments
A specification for potassium hydroxide is contained in the Food
Chemicals Codex (FCC).
The EINECS number for potassium hydroxide is 215-181-3.
Sumber : Handbook of Pharmaceutical Excipients; p; 606
key: monografi potasium hidroksida, potasium hidroksida adalah, potasium hidroksida,potassium hydroxide, potasium hidroksida hpe. hpe potassium hydroxide, potasium hidroksida, potassium hidrokside,